Processes leading to Element Concentration in Igneous Rocks
Similarity of many inter-element patterns makes it appear that the earth was originally formed of a mass of quite homogenous chondrite material consisting mainly of magnesium, silicon and iron. With the heat of compaction the native nickel-iron melted and worked it’s way down to form a metallic core. The remainder began a process still commonly seen of partially melting small quantities of basalt containing a much higher proportion of the elements which are not to any degree absorbed into olivine, orthopyroxene, clinpyroxene and plagioclase. Removal of these minerals from slowly cooling magma leads to higher concentration of these same element, the LILE, (Lithophile light elements) in the magmatic residua. So we now may find igneous rocks with up to 500 times the original chondrite levels of Cs, Rb, Ba, Th, U, Nb, K etc. Subaerial weathering extends these processes so than banded iron formation may contain 60% of Fe2O3, while other rocks may be pure calcium carbonate or pure quartzite (SiO2). Sulphur has the ability to combine with many metals including Cu, Ni, Zn, Co, Pb, Cd. Segregations of sulphides in igneous rock may have an average of several percent metal, making it much more available than the parts per million found in the parental chondrite.
Our whole civilization depends on finding and using these concentrations whether occurring as oxides, silicates, or sulphides and the processes must be understood; a great deal of research has gone into the origin of sulphides, less into silicates which are more refractory and more difficult to use as a source of metals. Even the nickel industry which obtains Ni from sources in peridotites and komatiites, relies on secondary Ni sulphides, not on the original Ni silicates. However as sulphide ore bodies become worked out, sources within silicates will become more important. In addition an understanding of the compositional envelope for the Oceanic Basalts which make up 60% of the Earth’s surface is dependent on an understanding of these differentiation processes.
Three of the more important elements used in differentiating between volcanic magmas are Zr, Nb and Y and can be used to illustrate processes. In the range of ORB and OIB rocks, Nb may vary in amount from 1 – 600 ppm, Zr from 38 – 2200 ppm and Y from 18 to 220 ppm. Even higher amounts especially of Nb are found in special rocks such as carbonatites. At present zirconium for example is recovered from zircon minerals in beach sands, Should these supplies run out, it is better we know that commendites have perhaps 20-30 times the amount of Zr than have some basalts. Other elements especially Cs, Rb, Ba, U, REE etc also show the manner in which elements may be concentrated and can be studied in the same manner as we shown here
 |
Zr/Nb, Y for the ORB parental magmas as seen in Macquarie Island, (Kamenetsky, 2000).
Zr increases linearly with the decreasing degree of partial melt. Y remains almost constant at 18 – 22 ppm while Nb increases very sharply from almost zero to 90 ppm with decreasing degree of melt.. It will be seen that different parental magmas will vary greatly in their Zr/Nb or Nb/Y ratio, in fact lavas may be found of Zr/Nb ranging from 30+ in NMORBs down to about 2.5 in very alkaline lavas. Fractionation by ol and or plag which contain little Y will push the Y content of low Zr rocks up very steeply, less so with high Zr magmas. Cumulates including clinopyroxenes,which take in up to 40-50 ppm Y, will elevate the residual magmas very little and may actually depress it. All three mineral ol, plag, cpx, exclude Nb almost entirely and will strongly elevate Nb in the residual magma |
Differentiation
Once formed, the parental basalt rarely reaches the surface unmodified. In the case of the depleted NMORBs, plagioclase and olivine form and may be separated from the magma which will have been depleted in Al, Sr, Mg, Ni and enriched in Na, Fe and the residual LILE including the REE and Nb. Neither plag nor ol contain more than infinitesimally small amounts of Zr, Y, Nb.
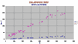 |
Zr/Nb, Y diagram for Galapagos Rise NMORBS. The parental magma was very low in NB so there has been little Nb enrichment. However the PM had about 20 ppm Y while ol + plag have virtually none. Removal of these two minerals has resulted in a massive build-up of Y to about 200 ppm as the rocks progressively changes from basalt to rhyolite. |
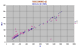 |
Zr/Nb,Y diagram for Socorro Id in the Revillagigedos. These rocks appear to be EMORBS a little more enriched than the Galapagos Ids. Being smaller degree partial melts, the parental magma had quite high Nb which has been greatly elevated by fractionation, whereas the Y has stayed at the same level as seen in the Galapagos Rise. Here Nb and Y have the same slope. These rocks are mildly alkaline with Zr/Nb of about 7 (Transitional = 10) |
Differentiation in OIB alkaline rocks
Here we are on less sure ground as we have few analysed glasses nor has anyone found a parental series as we have for the ORBs. However, they cannot be too different from the OIBs, being probably more magnesian and lower in silica but all are enriched in the LILE elements.
Fractionation does not commence with the formation of plag + ol but with cpx + ol. Cpx may include up to about 50 ppm Y, the Y increasing with the Ti content, so that the more alkaline rocks will become more Y depleted. Nb however is very low (sub ppm) in both cpx and ol so Nb builds up rapidly as we progress from alkali olivine basalt or basanite towards trachyte or phonolite. Alkaline rocks close to transitional separate a cpx of low Ti and Y content so Y still increases with fractionation.
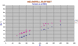 |
Zr/Nb, Y for the Helgafell-Surtsey series on Westamanneyjar Is, south of Iceland. |
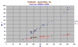 |
Zr/Nb,Y diagram for Tubuai, a highly sodic basanite nephelinite Island. No trachytes are present.
In non glassy whole rocks cumulates may distort the members of lower concentration. |
 |
Zr/Nb,Y for the Mauna Kea drill hole, Being tholeiitic Nb is less than Yb, Both elements trend towards zero with increasing olivine. |
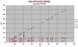 |
In this diagram we show the extreme depletion of Ba, Rb in the DMORBs of the Galapagos Rise. Not only is Ba less than Zr, it is much less than Y being in some samples at sub 5ppm levels. The low Sr is due to fractionation of plagioclase, we have little idea of the true levels of Sr in such rocks.
In OIBs such as Volcan Ecuador (see Galapagos Islands in OIB chapter) and in Ascension Id in the Atlantic, Ba = Zr. |
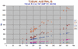 |
Zr diagram for Tubuai in the Austral Is., a highly sodic basanite-nephelinite series. Note the Ba elevated above Zr and the Rb > Y as well as the high Nb, (and Sr).
A few islands have higher Nb, eg, Gough and Bermuda.
The ORB and OIB seem to form a complete series as regards their LILE content, being completely gradational without breaks.
Claims of contamination by sediments or lithospheric rock do not seem possible. |
In all ORB –OIB lavas, Ba, Rb, Th U K, LREE, are progressively elevated relative to Zr in the same manner Nb is.